BAPETEN Conducts Parallel License Verification at Muhammadiyah Islamic Hospital Kendal and Ciputra Hospital Surabaya
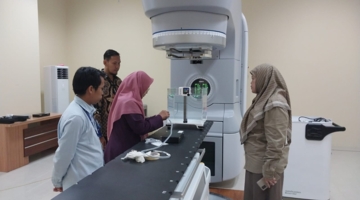
Kembali 02 Februari 2026 | Berita BAPETEN | 363 lihatBAPETEN, through the Directorate of Licensing of Radiation Facilities and Radioactive Materials (DPFRZR), conducted parallel verification activities for a radiotherapy facility at Muhammadiyah Islamic Hospital Kendal and a nuclear medicine facility at Ciputra Hospital Surabaya. The verification process was carried out from 2 to 5 February 2026, to ensure the safety and integrity of radiotherapy and nuclear medicine bunkers in order to protect the safety of workers, patients, the public, and the environment.
The licensing verification activity at Muhammadiyah Islamic Hospital Kendal was attended by the Activity Manager of the Healthcare Facility Licensing Functional Group, Herry Irawan, who also served as the head of the verification team, along with team members. Meanwhile, the inspection team at Ciputra Hospital Surabaya was led by Senior Radiation Supervisor of the Healthcare Facility Licensing Functional Group, Ahmad Maulana, together with team members. In Kendal, the inspection team verified a radiotherapy facility utilizing a LINAC unit, Shinva brand, model XHA 1400. Meanwhile, in Surabaya, the inspection team conducted verification of an in vivo diagnostic nuclear medicine facility using a PET/CT system, Siemens brand, model Biograph Trinion (CT64), with radionuclide sources F-18 and Ga-68. In addition, the verification also covered nuclear medicine therapy facilities utilizing radionuclide sources I-131, Sm-153, Lu-177, and P-32.
The scope of the on site inspection included a review of documentation and technical verification to ensure that the constructed facility design complies with regulatory requirements, including confirmation of the adequacy of radiation shielding wall thickness in the radiotherapy bunker. At the in vivo diagnostic nuclear medicine facility, inspections were conducted across various critical areas, including preparation rooms, radionuclide or radiopharmaceutical counting and storage rooms equipped with standard-compliant fume hoods, radiopharmaceutical administration rooms, imaging rooms, post administration patient monitoring rooms, decontamination rooms, temporary radioactive waste storage rooms along with their processing areas, dedicated patient waiting rooms, dedicated patient restrooms, and isolation inpatient rooms for nuclear medicine therapy patients.
In addition, the team ensured that the main and supporting equipment were functioning properly through radiation exposure measurements and dosimetry testing. The inspection included an evaluation of patient service workflows and personnel competence in operating equipment such as LINAC and PET/CT in accordance with applicable standards. The team also examined radiation protection equipment, radioactive source security systems, and the reliability of safety systems used to ensure operational safety.
The verification results showed that the facility design has complied with the required safety standards. This was demonstrated through the design calculations of the radiation shielding structure. In addition, the main and supporting equipment, human resource competencies, and operational procedures for radiotherapy using a Linear Accelerator (LINAC), nuclear medicine services using PET/CT, and nuclear medicine therapy were also declared to have met the requirements for radiation safety and source security. Nevertheless, the hospital has committed to addressing and completing the findings identified by BAPETEN within the specified timeframe. [DPFRZR/Dwiangesti/BHKK/HR]

























Komentar (0)